GENERAL INFORMATION
Nonossifying fibroma is a benign intracortical, multilocular and well circumscribed lesion that affects young patients. It originates from the proliferation of fibrous tissue and histocytes. The most common location are the distal femoral and distal tibial methaphyses. Children and adolescents are the most common group of age affected. The lesion may move into the diaphysis with bone growth. A non ossifying fibroma is a benign indolent tumor meaning that it does not actively grow and destroy bone however it may weaken the bone and predispose the bone to fracturing. At skeletal maturity most NOF begin to heal in on their own. In fact, many NOF are incidentally found later in the second decade of life, as a healed lesion. Nonossifying fibroma is usually asymptomatic. When non ossifying fibromas are less than 2 cm they are referred to as fibrous cortical defects.
CLINICAL DATA
• Most common tumor in children, and has been estimated that up to 30% of children will have this lesion.
•
> 50% of the cases are diagnosed in the first 3 decades of life
•
Discovered as an incidental finding, usually asymptomatic
•
Preference for male than in females (2:1)
•
Less than 5% of patients have multiple non ossifying fibromas and are associated with neurofibromatosis or Jaffe-Campanacci syndrome; Café-au-lait spots are associated with both of these conditions
•
Usually begin to heal after skeletal maturity and the healing process works its way from the area most distal from the growth plate toward the growth plate
•
In extremely rare instances an aneurysmal bone cyst (ABC) can arise from an NOF. The ABC will grow and destroy bone.
•
Fibrous Cortical Defect: Small lesion (up to 1cm) that involves only the cortex (Fig. 5-7)
Differential diagnosis
Benign lesions
• Chondromyxoid fibroma
• Fibrous dysplasia
• Benign fibrous hystocytoma
• Desmoplastic fibroma
• Giant cell tumor of bone
• Aneurysmal Bone Cyst
Malignant lesions
• Malignant fibrous histocytoma
• Osteosarcoma
CLINICAL PRESENTATION
Signs/Symptoms
• Sometimes physical examination is unrevealing
• Pathologic fracture may be the initial presentation.
• When is symptomatic, may present discrete swelling near the affected area.
• Cutaneous pigmentation (Café-au-lait spots) may be seen, and may be associated with multiple lesions
Prevalence
• Slight preference for Male>Female (1-2:1)
• Age of presentation is between 5 to 10 or 20 to 25 years.
• Peak age second decade of life.
• Extremely rare in adults
Sites
• Distal femur, distal tibia and proximal tibia count close to 80%.
• Less commonly found in upper extremity
• May also multifocal
• Arise in the metaphysis eccentrically arising from the cortex
RADIOGRAPHIC PRESENTATION
Plain x-ray
• Lytic and irregular; Well circumscribed lesion with surrounding sclerosis (fig 1-3).
• Lesions are metaphyseal and eccentric arising from or involving the cortex
• May appear expansile and sharply marginated with sclerotic rim
• The long axis of the lesion usually runs parallels to the affected bone
• Multiple lesions could be present
• New bone fills the defect and in some instances is denser than adjacent cortical bone leaving a residual “scar” on an xray, CT or MRI
• Often has reactive internal trabeculations
CT
• Useful for determining the true extent of the lesion
• Demonstrates well defined sclerotic borders
• Demonstrates expansile nature and scalloping of bone
• Useful for demonstrating internal trabeculations
MRI
• Low- signal on T1 (Fig. 8)
• Variable heterogeneneous signal on T2 (Fig. 9).
• Possible extensive hypercellular fibrous tissue and hemosiderin seen on T2
• Can be useful for demonstrating an aneurysmal bone cyst component
Whole body bone scan
• Sometimes useful to detect multiple lesions
• Usually low level of uptake on bone scan unless a fracture has occurred
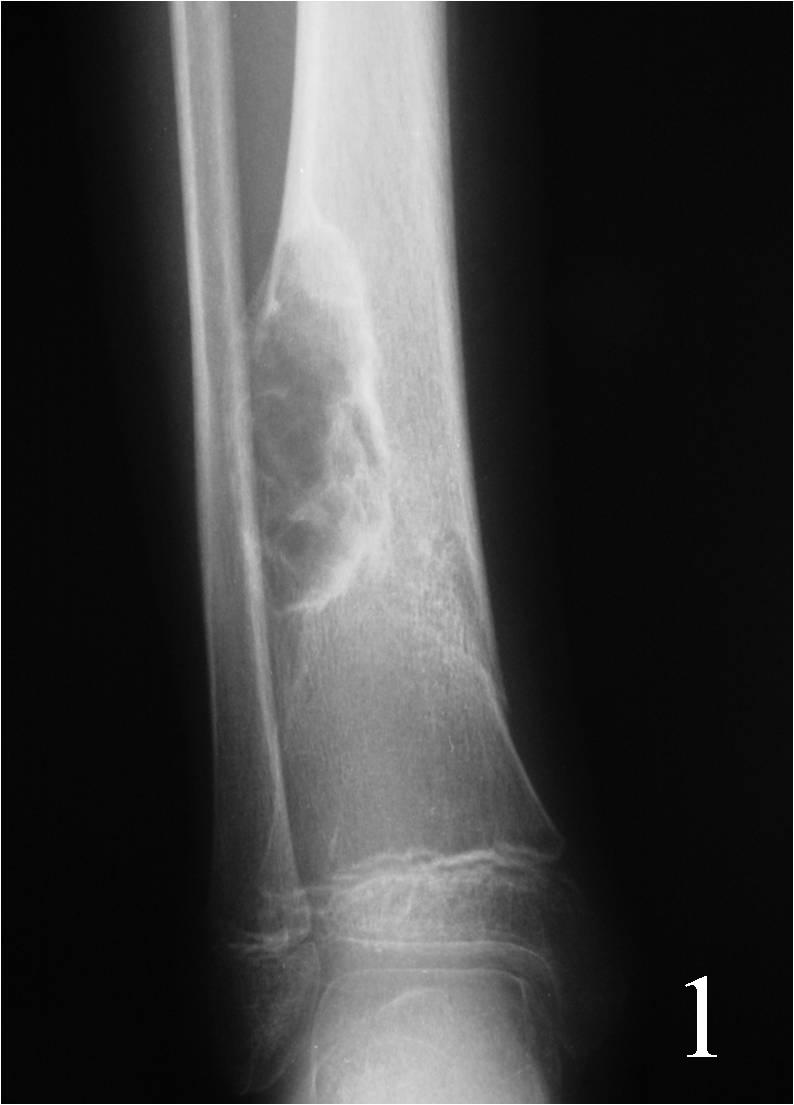 Fig. 1
Fig. 1
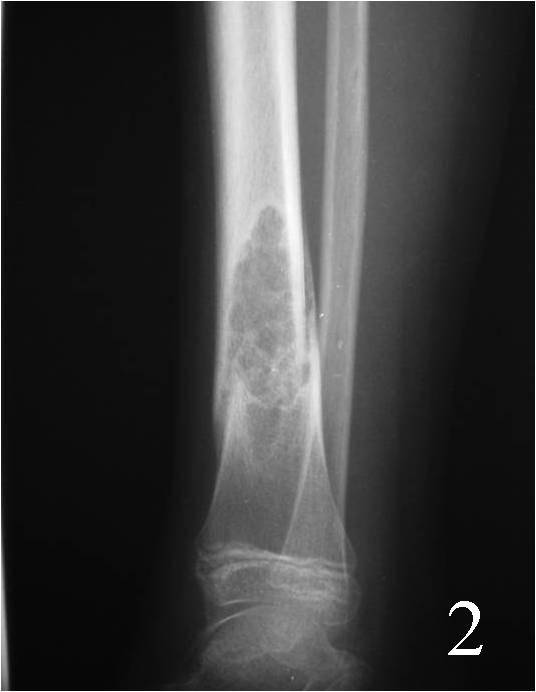 Fig. 2
Fig. 2
Fig. 1-2: AP (Fig. 1) and lat (Fig. 2) plain x-ray of the distal tibia-fibula shows a nonossifying fibroma of the distal tibia with a nondisplaced pathologic fracture. The lesion is geographic, well circumscribed, lytic, eccentric, arising in the metaphysis and has internal trabeculations. There is a sclerotic margin.
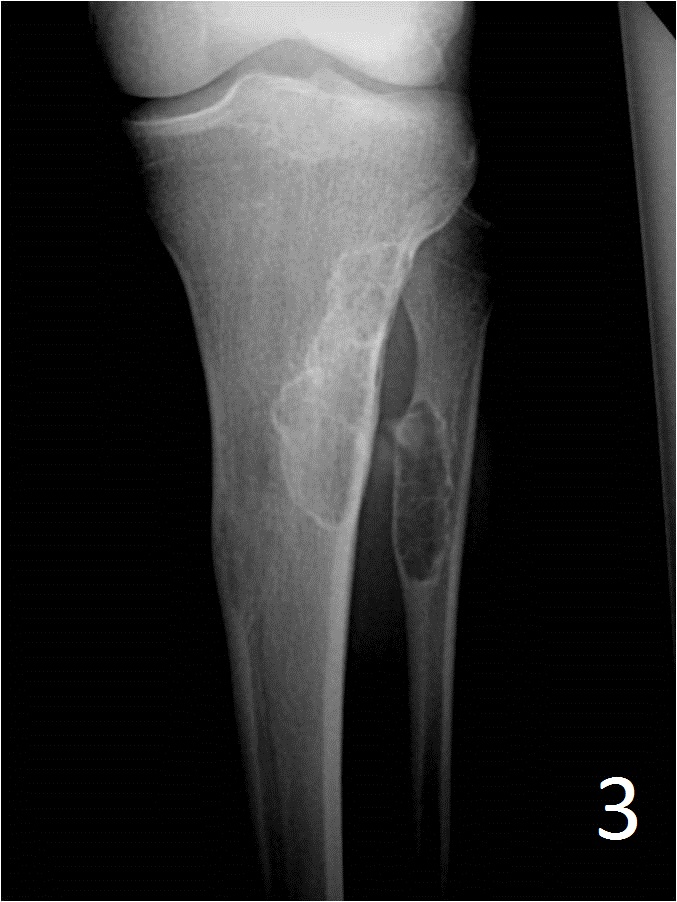 Fig. 3: AP xray of tibia-fibula shows a healed (ossified) NOF of the proximal tibia and fibula.
Fig. 3: AP xray of tibia-fibula shows a healed (ossified) NOF of the proximal tibia and fibula.
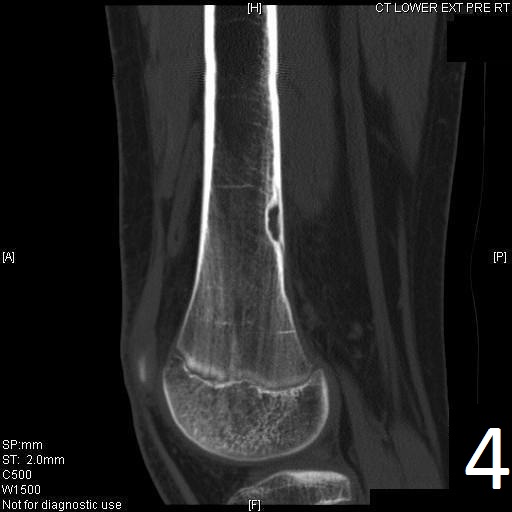 Fig. 4
Fig. 4
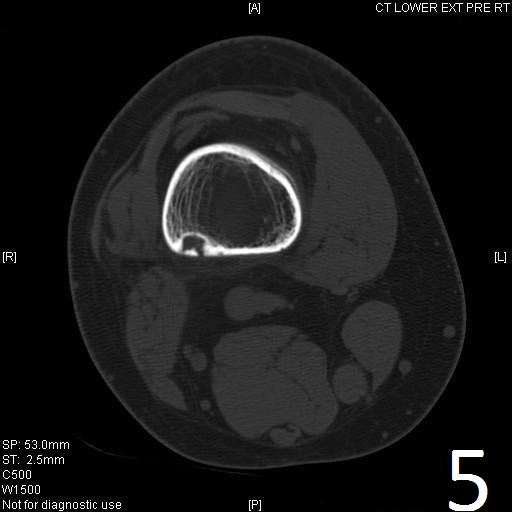
Fig. 5
Fig. 4-5: Sagittal (Fig. 4) and axial (Fig. 5) CT reconstruction of the lower extremity shows a well circumscribed, small sclerotic cortical lesion consistent with a fibrous cortical defect.
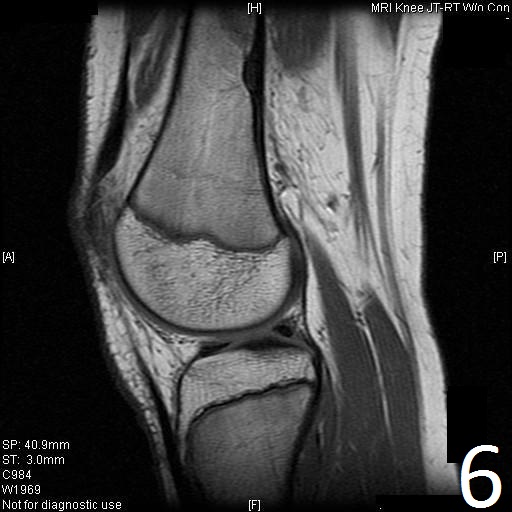
Fig. 6
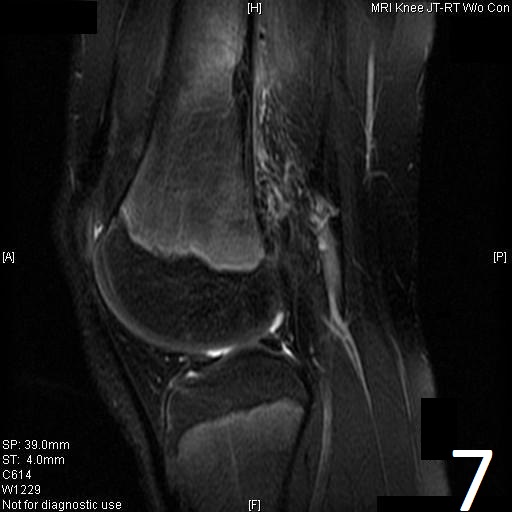
Fig. 7
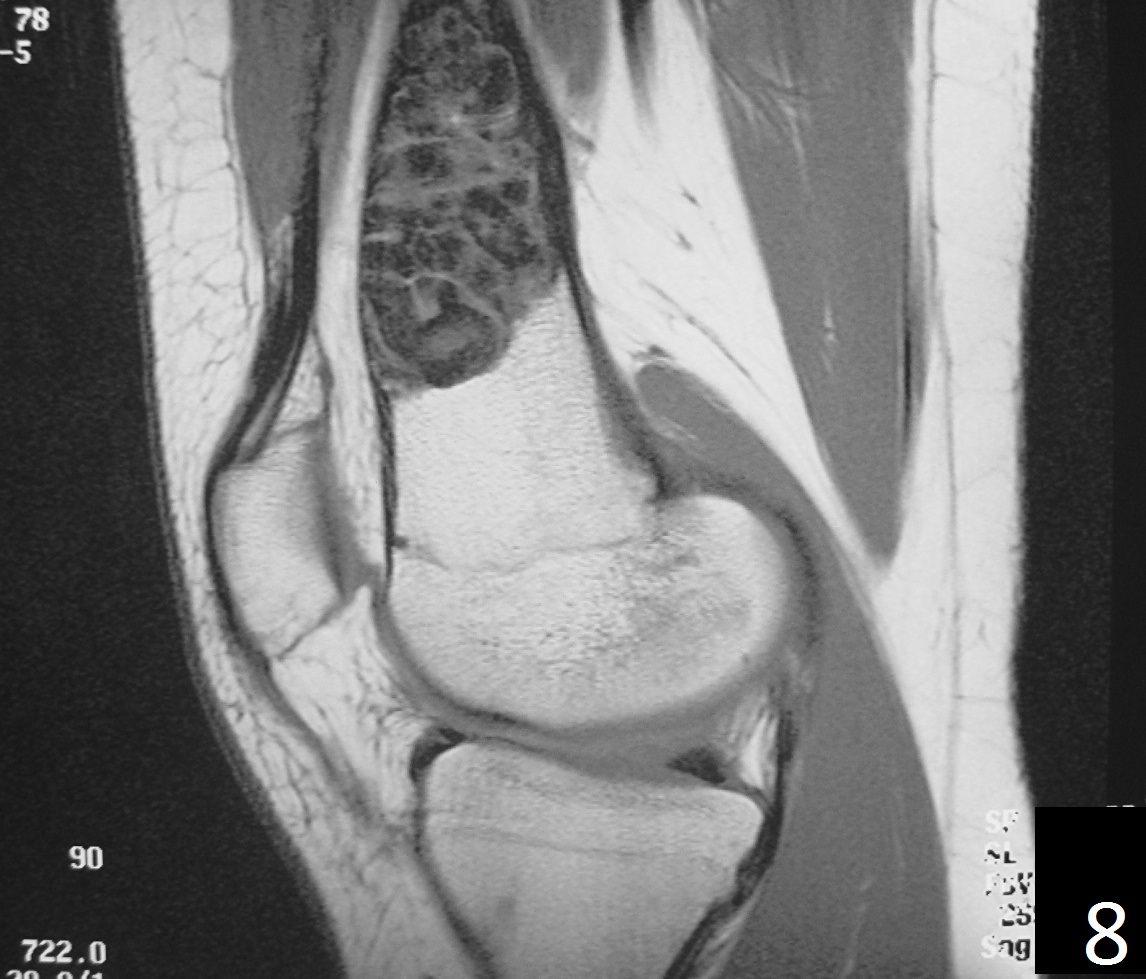
Fig. 8
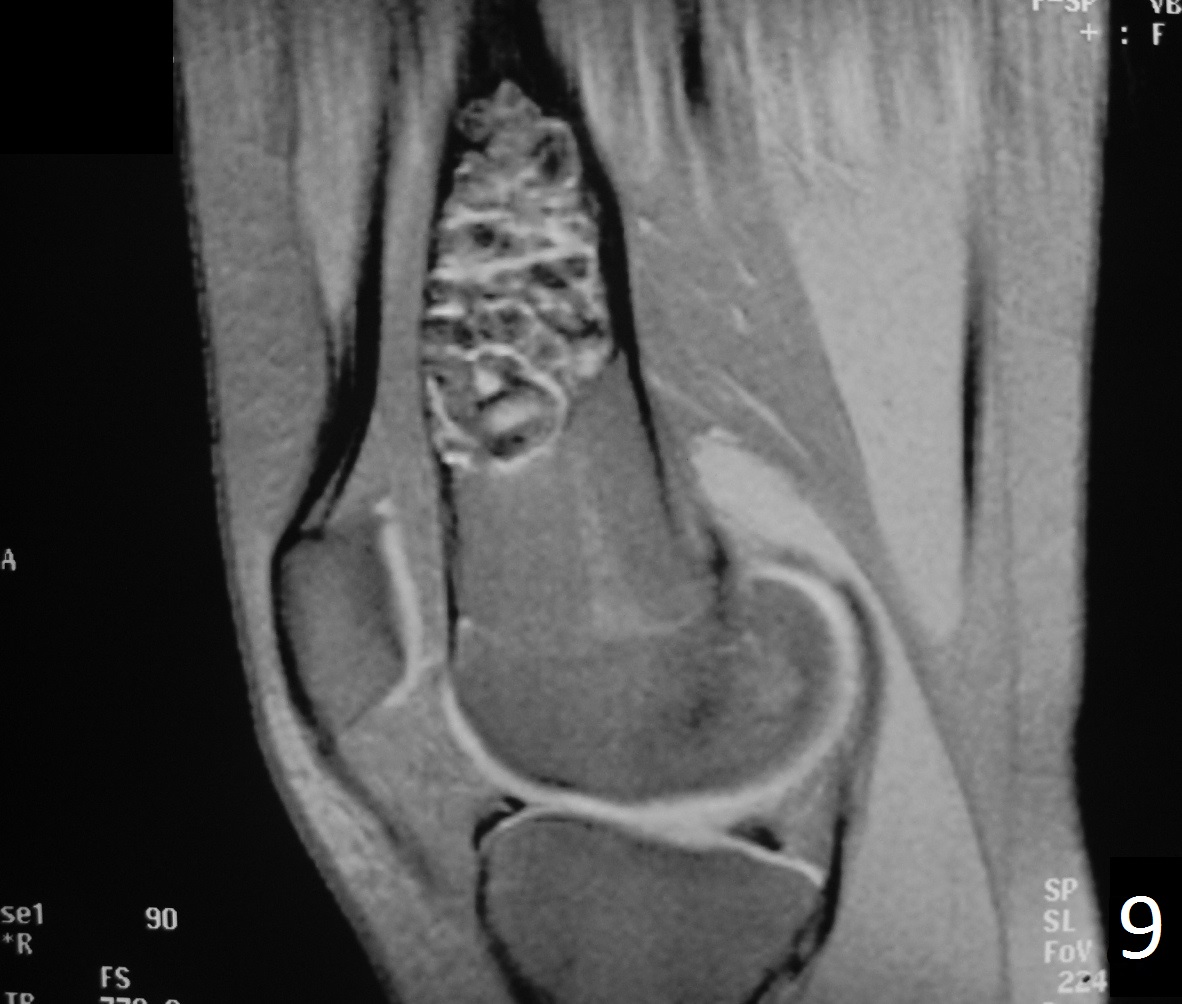
Fig. 9
Fig. 6-9: Sagittal T1 (Fig. 6) and T2 (Fig. 7) MRI of the knee demonstrates a fibrous cortical defect. Axial T1W (Fig. 8) and T2W (Fig. 9) show a heterogeneous lesion on distal femur, compatible with nonossifying fibroma.
PATHOLOGY
Gross
•
Curetted fragments are fibrous and vary from brown to yellow color.
Microscopic
•
Spindle cells are distributed in a storiform (swirling) pattern and mixed with other foci containing abundant histocytic cells (Fig. 10-11).
• Lipid laded histiocytes and macrophages
• Giant cells are present in irregular cluster
• Clusters of hemosiderin-iaden macrophages are dispersed
• The nuclei of the spindle cells and histocytes are regular
• Bland appearing cells with small uniform nuclei and no nuclear pleomorphism
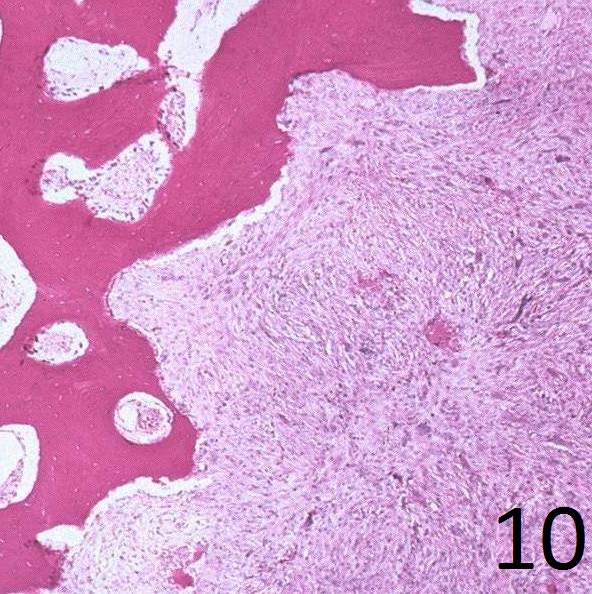
Fig. 10
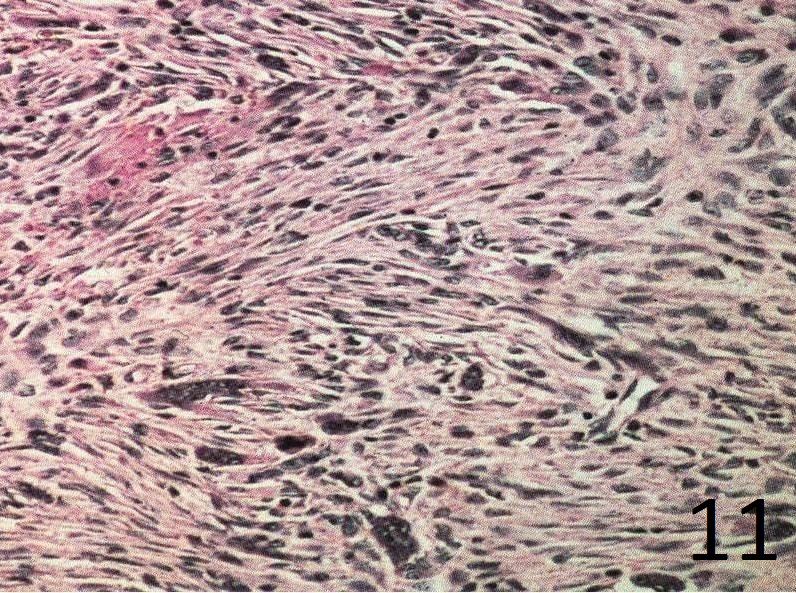
Fig. 11
Fig.10: Microscopy: Low power view shows a spindle cell tumor with fibrous stroma with the cells arranged in a storiform pattern. There is thickened bone at the periphery demonstrating that the lesion has a sclerotic margin.
Fig.11: High power view of the lesion shows the storiform patern, bland spindle cells, fibrous tissue and hemosiderin laden macrophages.
PROGNOSIS
Biological Behavior
• Benign tumor, indolent and not active nor aggressive.
• The tumor may be large enough to place the bone at risk for fracture (probably greater than 50% of the diameter of the bone in both directions).
• When the patient reaches skeletal the maturity, the lesions are prone to ossify (heal).
TREATMENT
• Observation indicated when the lesion does not threaten the integrity of the bone
• Surgical resection may be recommended when the lesion is more than 50% of the bone diameter. Surgery usually consists of a curettage and bone grafting.
• Curettage and bone grafting is curative when required
• In cases of the pathologic fractures consideration should be given to allowing the fracture to heal first then treating the nonossifying fibroma
